 
Placed separately at the bottom portion of the periodic table. Which period in the periodic table has both s The. (d) f - block elements : An element in which outermost (differentiating) electron of its atom enters to f - sub shell of anti- penultimate (n-2) shell is called f - block elements. They are : (i) s-block, (ii) p-block (iii) d- block and (iv) f-block. Each different element has atoms with a different and. Some examples of elements are gold, oxygen, neon, potassium, and tungsten. p-block of the periodic table has its valance electron in p-subshell. 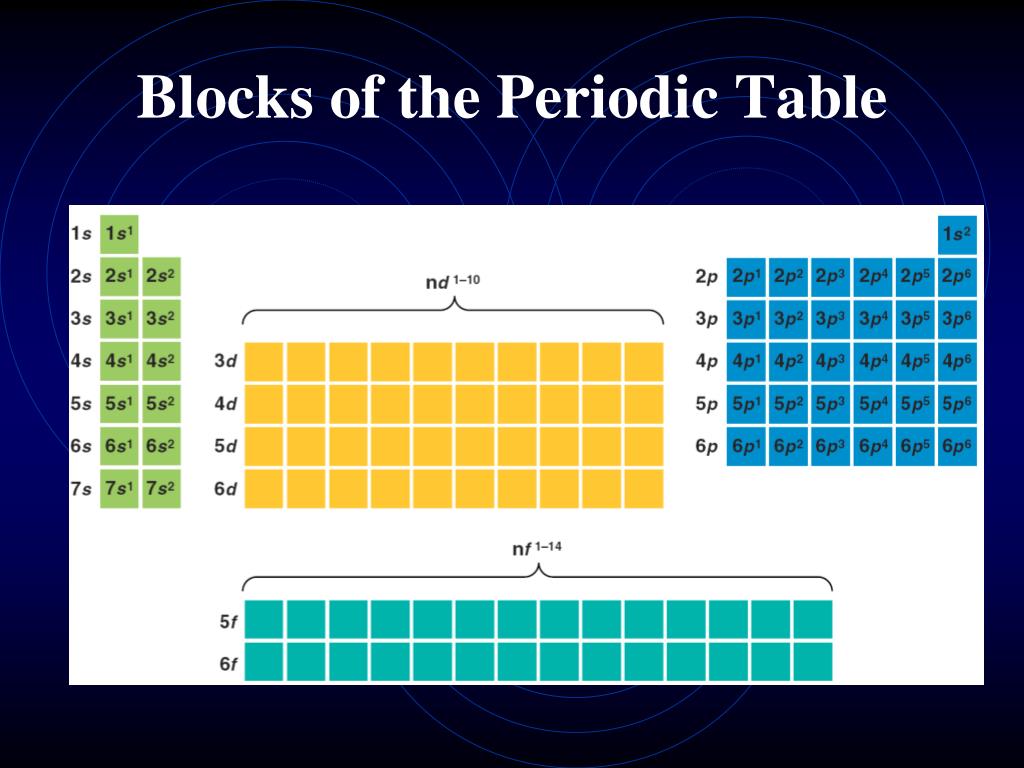
s-block of the periodic table has its valance electron in s-subshell. Kept at middle (between s and p block elements) portion of the periodic table, group 3 to 12. Each little block on the periodic table represents one element. The different blocks of the periodic table are s, p, d and f blocks. A cut between the halogens and the alkali-metals, where the largest change of chemical behavior occurs, became the most favored one, at least among practicing chemists. 26 February 2019 This reimagining of the periodic table, proposed by chemist Theodor Benfey in 1964, emphasises the continuity of the elements rather than imposing artificial breaks RUN your. (c) d - block elements : - The atom of an element in which outermost (differentiating) electron enters to d - sub shell of pen ultimate (n-1) shell is called d - block elements. Later inventors of Periodic Tables cut, so to speak, this cylindrical bent surface at different points, in order to obtain flat printable tables. General electronic configuration of p - block elements is n s 2 n p 1 − 6īoth s and p block together is called representative or normal elements. The names transition metals and inner transition metals are often used to refer to the. Kept in right hand side of the periodic table, group 13 to 18.Įlements in group 18 are called Aerogens or Noble gases. They are placed in a separate panel at the bottom of the periodic table. (a) s- block elements : An element in which the outermost (differentiating) electron of its atom belongs to s - orbital of valence shell is called s - block. The representative elements are those in which the distinguishing electron enter an s or p subshell. Each block indicates which electron sublevel is in the process of being filled. Should a new element be discovered, it will be in g-block. This is the first period of the periodic table that involves d-block elements, 3d sub-shell in the process of completion for d-block elements. This is a list of the 118 chemical elements that have been identified as of 2023. The four block names are s-block, p-block, d-block, and f-block. Periodic table blocks are sets of elements grouped by their valence electron orbitals. (b) p - block elements: An element in which the outermost (differentiating) electron of its atom belongs to p - orbital of valence shell is called p - block elements. The type of subshell ( s, p, d, f )into which the distinguishing electron is placed is very closely related to the chemical behavior of an element and gives rise to the classification shown by the color-coding on the periodic table seen here. They are determined by the valence electron orbital. Kept in left hand side of the periodic table, group I
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
